A New Grand Bargain to Improve the Antimicrobial Market for Human Health
Final Report of the Center for Global Development's Working Group

By Anthony McDonnell, Katherine Klemperer, Morgan Pincombe, Rachel Silverman Bonnifield, Prashant Yadav, and Javier Guzman

Every year, antimicrobial resistance (AMR) directly causes 1.27 million deaths and is associated with an additional 3.7 million deaths.
Low- and middle-income countries bear the brunt of this burden, accounting for nearly 90 percent of the direct death toll and over 99.5 percent of AMR–related deaths among children under five.
Already, more people die directly from AMR than from HIV/AIDS, malaria, or any one form of cancer other than lung cancer.
And the problem of AMR is only growing: Without effective antibiotics, it has been estimated that 10 million people a year could die from AMR—equivalent to the annual death toll from all cancers.
Some life-saving surgeries and treatments, including transplants, will not be possible because the risks from these procedures will be too high without effective antibiotics.
Tackling AMR requires global consensus and action on many fronts.


Creating an antimicrobial market that ensures adequate innovation, access, and stewardship is key to fighting AMR.
Improving infection prevention and control, increasing surveillance, and reducing antibiotic use in agriculture are also critical.
Progress has been made to garner political commitment to tackle AMR, including through high-level commitments at the 2015 World Health Assembly and the 2016 UN General Assembly.
More than 170 National Action Plans (NAPs) were developed to formalize government responses to AMR.
New R&D initiatives have been created (such as CARB-X, GARDP, and the AMR Action Fund), and some countries have started to pilot new ways to value and procure antibiotics, including the United Kingdom’s subscription model and Japan’s pilot for a revenue guarantee system.
Surveillance systems have been improved through the Fleming Fund, and better data for decision making are available via the R&D Hub.
These policies and commitments are laudable, but collective action problems still stand in the way of realizing a new international approach to the antimicrobial market.
Building on its previous research and analysis on AMR, in 2022, CGD launched the working group A New Grand Bargain to Improve the Antimicrobial Market for Human Health to examine policy options to drive global action against AMR.
We outline the principles of a Grand Bargain that we believe all stakeholders can and should sign up to during the UN General Assembly's High-Level Meeting on AMR in 2024.
Balancing antimicrobial innovation, access, and stewardship
The current market for antimicrobials fails to spur research and development (R&D), ensure access to new and existing products, and protect against overuse.
These three objectives—innovation, access, and stewardship—must be balanced to ensure sustainability and equity. Global commitment and action are needed to overcome collective action problems and realize a new market structure for antimicrobials.
Three factors needed for a sustainable antimicrobial market
Access
without stewardship, speeds resistance and undermines innovation
Stewardship
constrains access and undermines innovation without a balanced policy
Innovation
unjust without access, wasteful without stewardship




Innovation, Access, and Stewardship
Three factors needed for a sustainable antimicrobial market
Access
without stewardship, speeds resistance and undermines innovation
Stewardship
constrains access and undermines innovation without a balanced policy
Innovation
unjust without access, wasteful without stewardship
Inadequate innovation
The R&D pipeline for antimicrobials is—and for decades has been—dry.
Inadequate access
Inadequate access to existing and new antimicrobials is a leading cause of death from AMR infections and a key priority in the AMR response in LMICs.
Inadequate stewardship
The current market for antimicrobials contains structural failures and perverse incentives that undermine stewardship, contribute to higher levels of inappropriate use, and drive up resistance rates.
Six Recommendations
from CGD’s working group A New Grand Bargain to Improve the Antimicrobial Market for Human Health
This report presents recommendations on increasing the availability of critically needed drugs, creating incentives to develop new ones, and reducing market pressures to misuse or oversell the drugs.
It provides one political and five operational recommendations.
The political recommendation outlines why it is both possible and in everyone’s interest to overcome the collective action problems inherent in dealing with market failures in the antimicrobial market through a global agreement.
The five operational recommendations describe actions countries could take to begin to implement such a deal.
Overview of the proposed Grand Bargain to Improve the Antimicrobial Market for Human Health





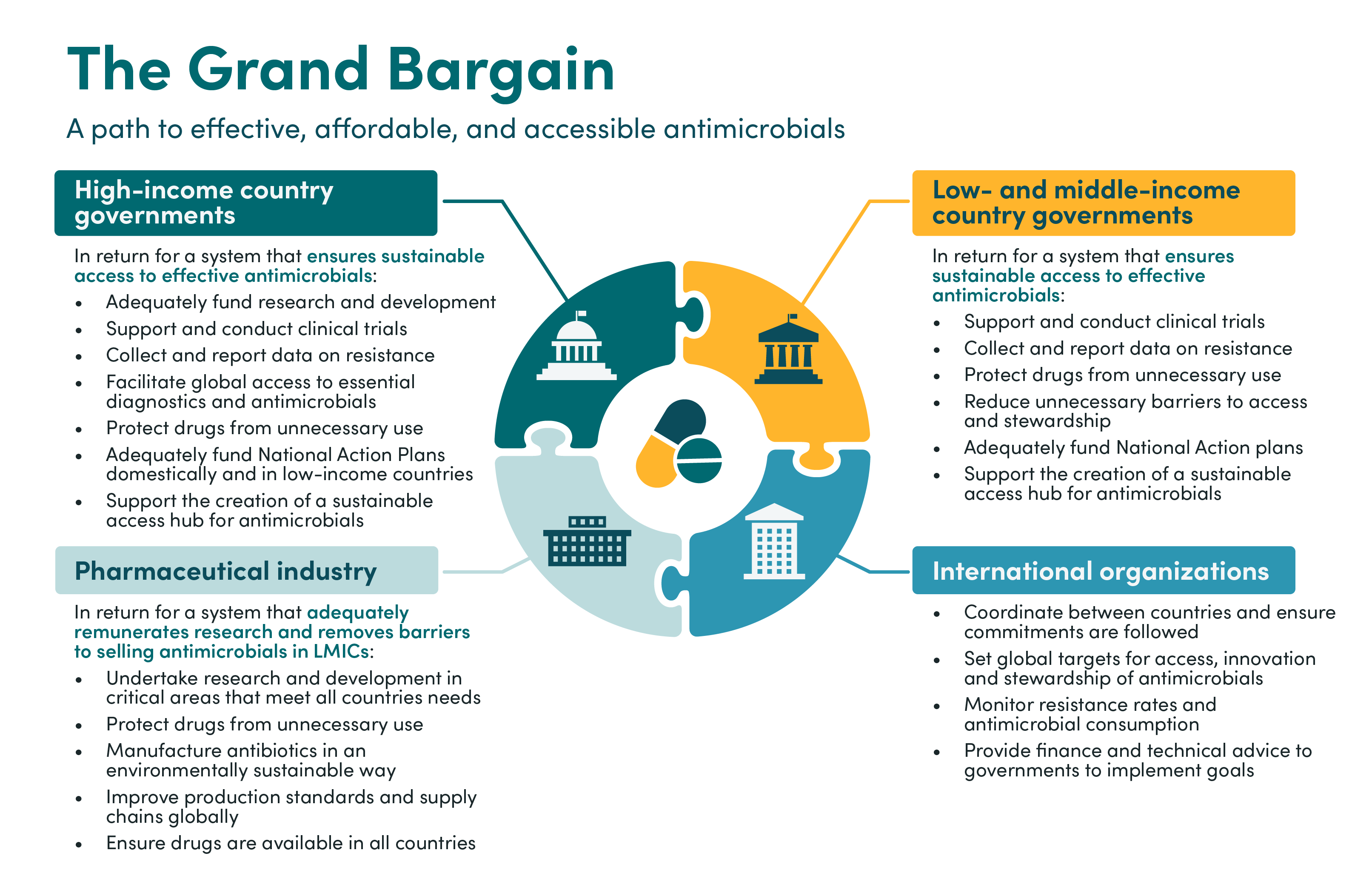
The Grand Bargain
A path to effective, affordable, and accessible antimicrobials
High-income country governments
In return for a system that ensures sustainable access to effective antimicrobials:
- Adequately fund research and development
- Support and conduct clinical trials
- Facilitate global access to essential diagnostics and antimicrobials
- Protect drugs from unnecessary use
- Adequately fund National Access Plans
- Domestically and in low-income countries
- Support the creation of a sustainable access hub for antimicrobials
Low- and middle-income country governments
In return for a system that ensures sustainable access to effective antimicrobials:
- Support and conduct clinical trials
- Collect and report data on resistance
- Protect drugs from unnecessary use
- Reduce unnecessary barriers to access and stewardship
- Adequately fund National Action plans
- Support the creation of a sustainable access hub for antimicrobials
International organizations
- Coordinate between countries and ensure commitments are followed
- Set global targets for access, innovation and stewardship of antimicrobials
- Monitor resistance rates and antimicrobial consumption
- Provide finance and technical advice to governments to implement goals
Pharmaceutical industry
In return for a system that adequately remunerates research and removes barriers to selling antimicrobials in LMICs:
- Undertake research and development in critical areas that meet all countries needs
- Protect drugs from unnecessary use
- Manufacture antibiotics in an environmentally sustainable way
- Improve production standards and supply chains globally
- Ensure drugs are available in all countries
A Sustainable Access Hub
that ensures reliable access to and stewardship of essential antimicrobials and diagnostics where the market currently fails
Manufacturers
Sustainable Access Hub
Patients
Benefits
Ensure access
Every year, antimicrobial resistance (AMR) directly causes 1.27 million deaths and is associated with an additional 3.7 million deaths.
Reduce inappropriate use of antimicrobials
by providing diagnostics, financial and technical assistance, and reducing incentives to oversell
Help countries track and report
consumption data to WHO's GLASS platform
Reduce shortages
by shaping markets and forecasting and aggregating demand, tracking suppliers' capacity and stockpiling where necessary
Reduce prices
of medicines through pooled procurement and multi-year contracts